Two infants had successful bone marrow transplantation with restoration of normal cellular and humoral immunity, but erythrocytic ADA deficiency persisted. None of the infants had detectable erythrocyte ADA activity. (1975) reported 3 affected infants from 2 families with SCID due to ADA deficiency inherited in an autosomal recessive pattern. The finding that both pairs of parents had an intermediate level of red cell ADA supported recessive inheritance the parents of the first child had about a 50% level of normal, whereas the parents of the second child had about a 66% level. The parents of the first child were related and the second child had a sister who died as a result of a major immunologic defect ( Hong et al., 1970). Mild upper respiratory infections began at age 24 months and progressed to severe pulmonary insufficiency and hepatosplenomegaly by age 30 months.
The other, aged 3.5 years, was allegedly normal in the first 2 years of life. One child, aged 22 months, had recurrent respiratory infections, candidiasis, and marked lymphopenia from birth. (1972) reported 2 unrelated girls with impaired cellular immunity and absence of red cell adenosine deaminase activity. Severe combined immunodeficiency, X-linked Severe combined immunodeficiency due to ADA deficiency SCID, autosomal recessive, T-negative/B-positive typeīare lymphocyte syndrome, type II, complementation group B Severe combined immunodeficiency, B cell-negativeīare lymphocyte syndrome, type II, complementation group Dīare lymphocyte syndrome, type II, complementation group A Severe combined immunodeficiency, Athabascan type Severe combined immunodeficiency with microcephaly, growth retardation, and sensitivity to ionizing radiation To learn more about the ADA-SCID gene therapy research and for a more in-depth explanation of the gene therapy process, read this news release from the National Institutes of Health.Bare lymphocyte syndrome, type II, complementation group Eīare lymphocyte syndrome, type II, complementation group C “Importantly, gene therapy is a one-time procedure that offers patients the hope of developing a completely functional immune system and the chance to live a full, healthy life.”

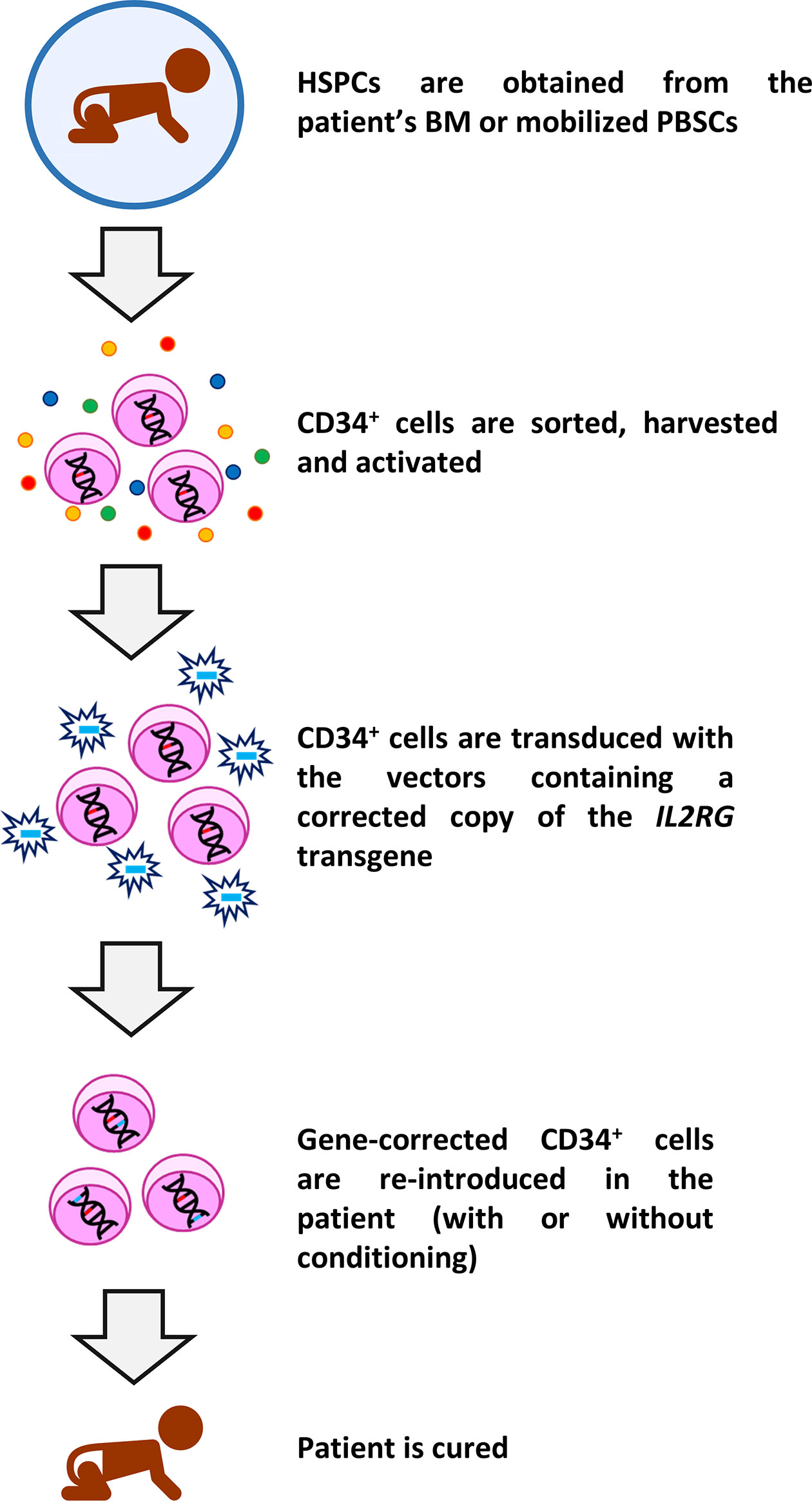
Fauci, M.D., director of NIH’s National Institute of Allergy and Infectious Diseases (NIAID). “These findings suggest that this experimental gene therapy could serve as a potential treatment option for infants and older children with ADA-SCID,” said Anthony S. Prior gene therapy trials for ADA-SCID used a gamma retrovirus to transfer the gene into the stem cells, resulting in leukemia in some patients. The virus, or vector, is different from the one researchers used in past gene therapy clinical trials for ADA-SCID. The absence of this enzyme leads to an accumulation of toxic metabolic by-products within lymphocytes that cause infection-fighting cells to die.įor these most recent clinical trials, researchers used a modified lentivirus to deliver a corrected copy of the ADA gene into the patients’ stem cells. The research study took place in both the United States and the United Kingdom.Ĭhildren with ADA-SCID lack an important enzyme called adenosine deaminase (ADA), essential for the metabolic function of a variety of cells, especially T cells. The researchers, funded in part by the National Institutes of Health, found that 48 out of 50 infants and children with ADA-SCID treated with gene therapy experienced a restored immune system within a period of two to three years and did not require additional treatments. Research findings recently published in the New England Journal of Medicine provide hope that gene therapy may one day be an accepted treatment for ADA-SCID instead of an experimental one.īabies born with severe combined immunodeficiency, or SCID, have no immune system and die from infection within the first year or two of life unless they undergo treatment to restore their immune system.
